7-7-7: 39% (78)
6-6-6: 35% (69)
7-6-5 : 9.5% (19)
7-6-6: 6.5% (13)
Don't care, just nerf it: 4.5% (9)
Don't care, just buff it: 3.5% (7)
8-8-8 : 2% (4)
What damage do you think the lightning gun should have?
180807 Hits


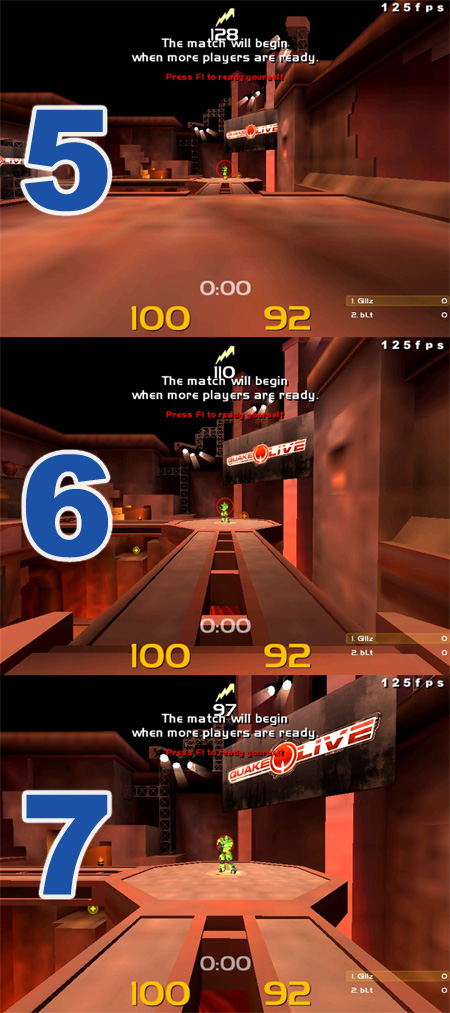 For those wondering the distance required to qualify for each damage amount, I made an image displaying the distances. It's not 100% accurate, each one will only be off by a VERY small amount of units.
For those wondering the distance required to qualify for each damage amount, I made an image displaying the distances. It's not 100% accurate, each one will only be off by a VERY small amount of units.